 Moreover, antibodies against vaccine toxoids/antigens persisted for up to a mean of approximately 6 years after booster vaccination, and the vaccine induced long-term immune memory against hepatitis B surface antigen and Hib antigen. In large clinical studies, Infanrix hexa elicited a strong immune response against vaccine toxoids/antigens, as indicated by high seroprotection/seropositivity/vaccine response rates and geometric mean titres.  Its immunogenicity and safety profiles were generally similar to those of currently available vaccines, the diphtheria, tetanus and acellular pertussis-based pentavalent vaccines plus monovalent HBV or Hib vaccines. Infanrix hexa as primary and booster vaccination was safe and highly immunogenic for all its component toxoids/antigens in infants aged <2 years, regardless of vaccination schedules. This article reviews the immunogenicity and protective effectiveness, as well as the reactogenicity and safety of Infanrix hexa. Infanrix hexa should be administered as a two- or three-dose primary vaccination course in infants aged 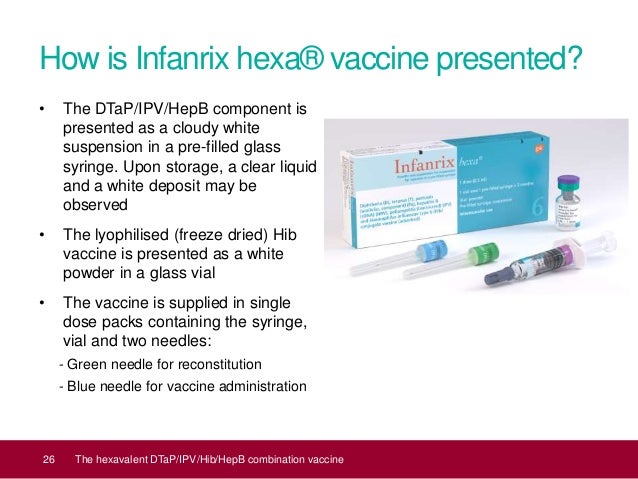 Infanrix hexa, administered intramuscularly, is a diphtheria, tetanus, acellular pertussis, hepatitis B (HBV), inactivated poliomyelitis and Haemophilus influenzae type b (Hib) conjugate vaccine, indicated for primary and booster vaccination of infants. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
